
by Cleanroom Connect
The implementation of EU GMP Annex 1 marks a transformative shift for the sterile filling industry. With new requirements emphasizing barrier technology and “first air” contamination control, pharmaceutical manufacturers face critical decisions in adapting their processes. Johannes Rauschnabel of Syntegon explores how equipment suppliers can play a pivotal role in facilitating compliance and seamless integration of these advanced standards.

by EdgeX, Inc.
EdgeX, Inc. announced today the commercial launch of two applications for environmental monitoring. Sense8 monitors and records temperature, humidity and pressure differentials to ensure regulatory compliance for operating rooms and pharmacy cleanrooms. Visual USP provides documentation of required tasks and staff competencies for USP 797/800 compliance.

by Dietrich Engineering Consultants
Dec Group, a leading provider of materials handling and process solutions, has announced a strategic partnership with IKA, a prominent leader in technology-driven solutions. This collaboration aims to revolutionize the battery manufacturing sector by offering comprehensive solutions for conveying and bulk handling, blending/dispersion, drying, mixing, milling, and packaging battery powders.

by LG Chem
LG Chem is set to establish a $3 billion battery cathode factory for electric vehicles (EVs) in Tennessee. Additionally, it has inked a long-term cathode material supply contract with General Motors (GM) worth a staggering $19 billion.

by RTX
Pratt & Whitney announced a $20 million investment to grow its manufacturing capacity in Singapore for the production of Pratt & Whitney GTF™ engine high pressure turbine (HPT) disks.
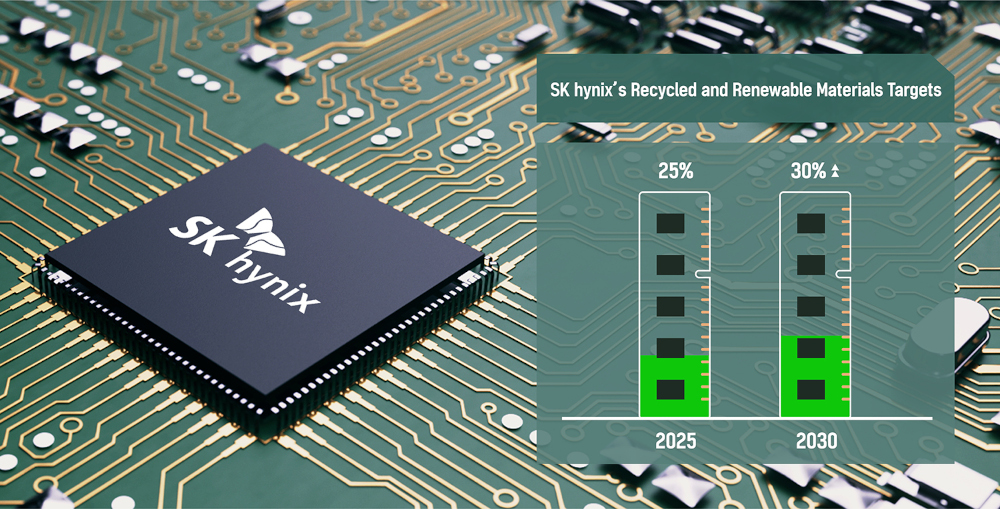
by SK hynix Inc.
SK hynix Inc. announced today that it has established a roadmap to actively utilize recycled1 and renewable2 materials in production, marking the first case that a semiconductor company lays out such mid- to long-term plan.

by BAE Systems, Inc.
Biologics Modular today announced a significant milestone with respect to intellectual property, demonstrating its ongoing commitment to innovation and leadership of the modular cleanroom industry. The patent award encompasses the area where cleanroom modules are preassembled with air filtration systems already in place. Therefore, when transported to the customer, it arrives as a pretested system, whether it is one unit or a cleanroom made up of multiple units connected togethe

by Toyota Motor North America
Toyota Advances its Commitment to vehicle electrification, Toyota will assemble three-row, battery electric SUV at Toyota Kentucky starting in 2025

by ImmPACT Bio
ImmPACT Bio Announces California-Based GMP Facility for Clinical Production of IMPT-314, a CD19/CD20 CAR T Therapy for B-Cell Malignancies and Autoimmune Diseases

by Biomerics LLC
Biomerics, the leading vertically integrated contract manufacturer for the interventional medical device market, announced today that it has entered into definitive agreements to merge with Precision Concepts’ medical business (“Precision Concepts Medical”).

by Spartech
Spartech, a leading manufacturer of engineered thermoplastics and custom packaging solutions, is pleased to announce that Royalite G50, a non-PVC rigid sheet product, meets stringent ASTM E84 Steiner Tunnel Test in accordance with their flame spread and smoke-developed indexes.

by UL Solutions
UL Solutions, a global leader in applied safety science, announced today that it plans to open a new battery testing laboratory in Auburn Hills, Michigan, in mid-2024












